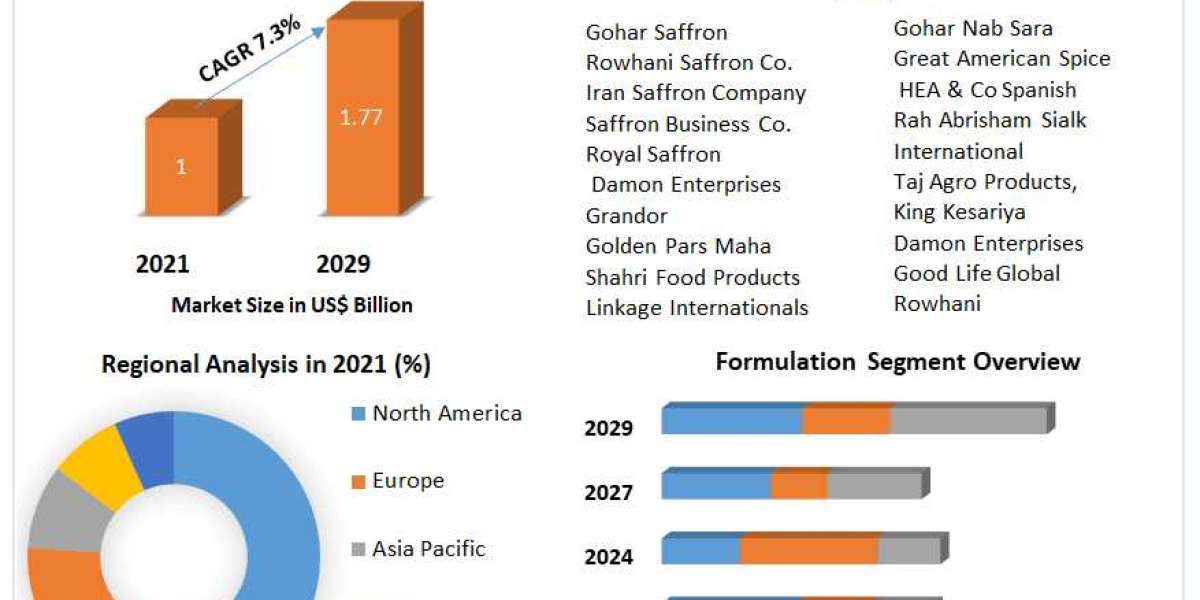
a harmonized positive list of monomers and starting materials for the manufacture of plastic materials and articles and a partial list of additives for plastic materials; Where appropriate, the lists contain specific restrictions (mainly specific migration limits (SML), maximum residual levels in material (QM), use restrictions, etc.). It also includes lists of substances that can be provisionally used at national level pending evaluation and final EFSA opinion. This guideline also sets an OML of 60 mg substances/kg food or food simulant or 10 mg/dm2 FCM surface area for any substance leaching from a material into food. Finally, the directive contains procedures for updating and completing the lists of authorized substances of azodicarbonamide (EU, 2008d).
Since July 2009, Directive 2002/72/EC has been amended by (EU, 2008d, e):
•
Directive 2004/1/EC suspending the use of the propellant azodicarbonamide from August 2, 2005.
•
Directive 2004/19/EC (EU, 2004b) which updates the positive lists of authorized substances and stipulates that the list of authorized additives becomes a positive list, with certain deadlines being set.
•
Directive 2005/79/EC (EU, 2005a) updating the positive lists of authorized substances.
•
Directive 2007/19/EC which establishes requirements for phthalates in gaskets in lids and introduces a grease consumption reduction factor to be considered in the migration tests and the functional barrier concept. In addition, the positive lists of permitted substances are updated and the ban on the use of azodicarbonamide in the manufacture of expanded plastic materials and articles is confirmed.
•
Directive 2008/39/EC updating the list of authorized substances used in the manufacture of plastic materials and articles intended to come into contact with food and the criteria for removing an additive from the preliminary list clarifies. It also stipulates that the EU list of additives will become a positive list from 1 January 2010. This means that from this date only the additives listed will be allowed for the manufacture of plastic FCMs in the EU. However, subject to national legislation, substances on the provisional list may continue to be used after that date, pending a final decision on their possible inclusion in the positive list of additives. This indicative list includes all additives currently being evaluated by EFSA and for which a petition was submitted in December 2006.
Search
Popular Posts
Categories
- Animals & Pets
- Antiques & Collectibles
- Art & Photography
- Auto & Cycles
- Betting Systems
- Books
- Business & Finance
- Children
- Computers / Internet
- Cooking, Food & Beverage
- Crafts
- E-Business & E-Marketing
- Education
- Electronics
- Employment & Jobs
- Enrichment
- Entertainment
- Ethnic
- Fashion & Style
- Fiction
- Games
- Green Products
- Health & Fitness
- Hobbies
- Home & Garden
- Languages
- Lifestyle
- Medical
- Men
- Mobile
- Music
- News & Politics
- Parenting & Families
- Reference
- Religion
- Science & Nature
- Self-Help
- Software & Services
- Spirituality, New Age & Alternative Beliefs
- Sports
- Supplements
- Travel
- United States
- Women
- Sponsored
- Other